
For the remaining two study periods, subjects received IBU Na tablets 2 × 256 mg or IBU LG 2 × 200 mg within 20 min of a standardized high-fat breakfast. In Study 1, conducted from July 29 to August 7, 2009, at PPD Development, LP (Austin, TX, USA), subjects received ibuprofen sodium dihydrate tablets 2 × 256 mg, solubilized IBU liquid capsules 2 × 200 mg, and standard IBU tablets 2 × 200 mg in each of three study periods following an overnight fast. Subjects remained on site for the duration of each respective study. In both studies, subjects received all interventions according to a computer-generated random sequence provided by Pfizer Consumer Healthcare’s Biostatistics Department treatment periods were separated by a washout period of at least 48 h. Two single-dose, randomized, open-label, inpatient, five-way crossover bioequivalence studies (Study 1, Study 2) were conducted to evaluate the pharmacokinetics of 400-mg dose equivalents of IBU administered in various different formulations. This report details two studies evaluating the pharmacokinetic profile of this new formulation in comparison with both standard IBU tablets and rapidly absorbed IBU formulations. Recently, a novel tablet formulation of IBU sodium dihydrate has been developed that has a thin-film coating and is manufactured using a patent-pending process. Clinically, IBU Arg has been found to provide a faster onset of pain relief versus standard IBU, while both IBU Lys and solubilized IBU have demonstrated more rapid pain relief compared with acetaminophen. Such formulations include IBU salt conjugates and gelatin capsules containing solubilized IBU that achieve maximum plasma concentrations ( C max) that are higher and are reached earlier (time to maximum measured plasma concentration T max) than those found with standard IBU tablets. 
In an effort to provide more rapid pain relief, newer IBU formulations have been designed to dissolve more readily in the acidic environment of the stomach. Patients experiencing acute painful conditions such as headache, musculoskeletal pain, menstrual cramps, or dental pain would benefit from a faster onset of pain relief. As a result, meaningful pain relief typically takes approximately 45 min after ingestion of an over-the-counter (OTC) dose (400 mg) of standard IBU. IBU, which is a carboxylic acid, shows low solubility in aqueous acidic media such as that which is found in the stomach. Although IBU is almost completely absorbed, allowing for nearly 100 % bioavailability, the rate of absorption depends on dissolution of the given formulation. With ibuprofen (IBU), one of the most widely used nonprescription analgesics available, pain relief is directly related to IBU plasma levels. When treating acute pain, rapid onset of relief is desirable. IBU Na is absorbed faster but to a similar extent as standard IBU formulations. Food slowed absorption of IBU Na and IBU LG similarly. After fasting, median IBU Na T max was comparable to that for IBU LG, IBU FG, and IBU Lys, but was much shorter than that for IBU Mot, IBU Nur, and IBU Adv. After fasting, AUC L was bioequivalent for IBU Na and IBU Mot, IBU Adv, and IBU Nur, but C max occurred significantly earlier with IBU Na. IBU Na was bioequivalent to IBU LG (fasted and fed) and IBU FG and IBU Lys (fasted) for rate ( C max) and extent (AUC L) of IBU absorption. Log-transformed area under the plasma concentration versus time curve to last observable concentration (AUC L) and maximum plasma concentration ( C max) were the primary pharmacokinetic parameters time to maximum measured plasma concentration ( T max) was analyzed post hoc. Study 2: In five study periods, fasted subjects received 400-mg IBU dose equivalents as IBU Na 2 × 256 mg, Advil ® FastGel ® (IBU FG) 2 × 200 mg, Nurofen ® (IBU Nur) 2 × 200 mg, Advil ® (IBU Adv) 2 × 200 mg, and Nurofen ® Express containing IBU lysinate (IBU Lys) 2 × 342 mg. In two periods following a high-fat breakfast, subjects received 400-mg IBU dose equivalents as IBU Na 2 × 256 mg and IBU LG 2 × 200 mg. 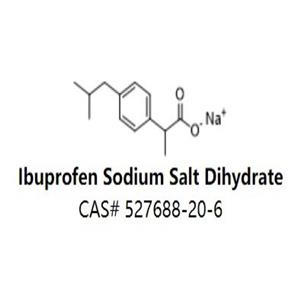
Study 1: In three periods, fasted subjects received 400-mg IBU dose equivalents as IBU Na 2 × 256 mg, Advil ® Liqui-Gels ® (IBU LG) 2 × 200 mg, and Motrin ® IB (IBU Mot) 2 × 200 mg tablets. Two randomized, single-dose, open-label, five-way crossover pharmacokinetic studies. Pharmacokinetic comparison of IBU Na versus other IBU formulations. A novel ibuprofen (IBU) formulation, Advil ® Film-Coated Tablets (IBU Na), was developed.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
